Clinical Mycobacteriology and Epidemiology (CLIME) Group
Projects

Selected concise host transcriptional signatures for the blood-based diagnosis of active tuberculosis in an HIV-prevalent setting (RNA-based diagnosis of TB; RADIANT)
This project (TMA2020CDF-3209 - RADIANT) is part of the EDCTP2 program supported by the European Union
Abstract
Tuberculosis (TB) is the biggest infectious cause of death worldwide, and the biggest cause of death in Sub-Saharan Africa among HIV-positive patients. There is need for a non-sputum-based rapid triage test that identifies individuals with presumptive TB requiring confirmatory diagnostic investigation. Such a test could reduce the burden on health systems, expedite referral and confirmatory testing, and treatment thereby reducing transmission.
A non-sputum triage test is needed as many symptomatic patients including those with HIV, can often not produce high quality sputum (which most current diagnostics rely on). Several blood transcriptional diagnostic signatures produced due to immune responses to M. tuberculosis infection have previously been described, however there is lack of real-world performance data especially in high TB/HIV-endemic African settings where rates of HIV (that could compromise sensitivity) and previous TB (that could compromise specificity) are high. Furthermore, by building on prior research that used untargeted sequencing approaches to identify candidate signatures, we are now at a stage to perform the targeted signature measurement at a large scale and cost-efficient manner as part of prospective diagnostic accuracy analyses in real-world settings.
Using the framework provided by an EDCTP-funded parent study (SeroSelectTB; PI Holm-Hansen), which is a pan-African evaluation of a point-of-care serological test for active TB, the candidate has a unique opportunity to pursue several important research questions. RADIANT aims are to 1) evaluate the sensitivity and specificity of selected concise peripheral host transcriptional signatures for active TB among symptomatic persons (in South Africa=1260 and Tanzania=840); 2) design a cost-optimised diagnostic algorithm based on transcriptional signatures, SeroSelectTB results, and confirmatory bacteriological testing, and 3) characterise bacteriologically-negative patients classified as non-TB to determine if those with elevated host transcriptional signatures (n=100 expected) have other respiratory pathogens (detected in nasopharyngeal swabs using a commercial multiplex panel) and/or develop active TB within six months (incident active TB).
The application includes 1) fellowship support for the candidate, 2) support for scientific and mentorship skills development (including support for a PhD student), 3) enhancement of capacity building and TB diagnostics expertise (specifically, the ability to evaluate next-generation biosignatures in clinical trials) to assist in transitioning the candidate to be an independent researcher with Africa-wide experience, and 4) the investigation of key research questions important for the next-generation of TB diagnostic tests. Hence, the candidate will not only lead research of immediate public health importance, but also grow as a young scientist.
Overarching goal:
To evaluate the diagnostic accuracy of selected concise host transcriptional signatures for the non-sputum-based diagnosis of active TB in symptomatic patients investigated in Africa and characterise patients who have elevated signature readouts but do not have active TB.
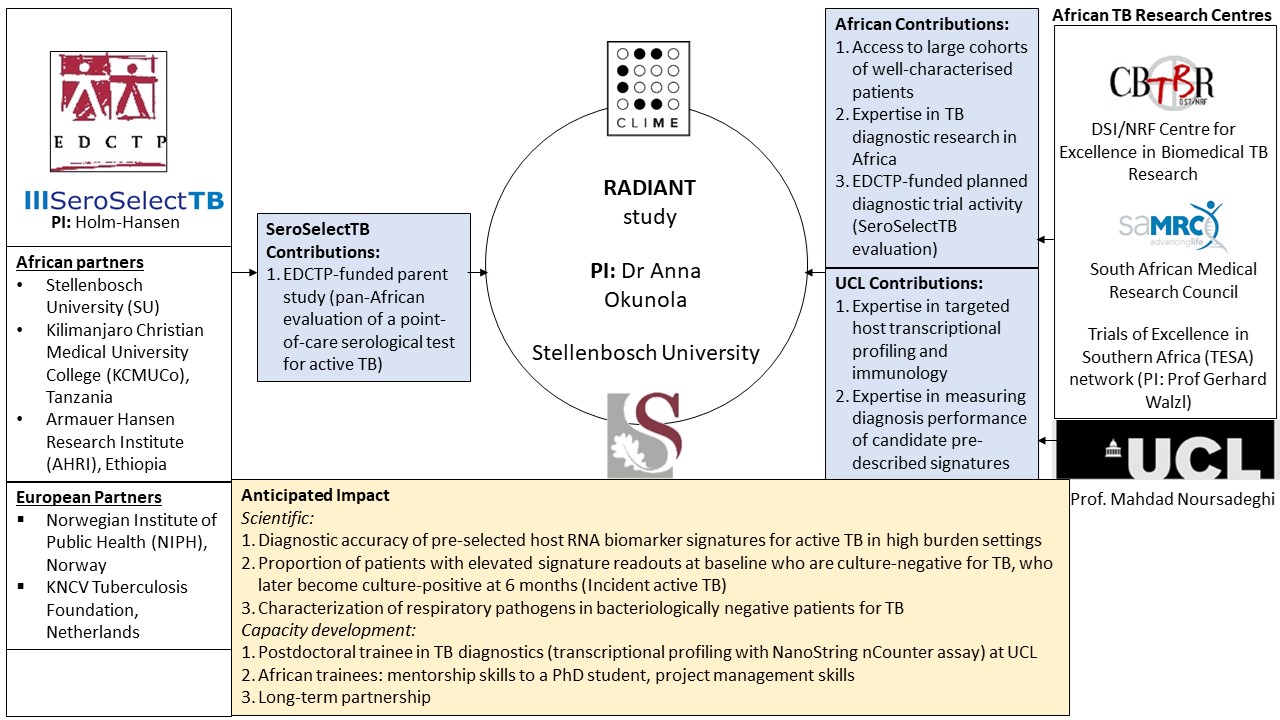
Figure 1. Networking and collaboration diagram showing the involvement of different institutions associated with the RADIANT study.
If you are interested in this project, please contact the PI (Dr Anna Okunola,
annaojo@sun.ac.za). Outputs from this project are acknowledged and reported alongside others from the CLIME group
here.

